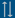MXY0291 Pichia pastoris is a genetically modified strain of the parental strain Bg11 (MXY0051) and was generated by a series of transformations.
The heme biosynthesis was first upregulated in Pichia pastoris through the sequential transformation with two vectors (pMX346 and pMX349) containing extra copies of 8 native P. pastoris heme biosynthetic enzymes driven by the strong native alcohol oxidase 1 promoter (P-aox1-PICPA). This increases intracellular concentration of heme to generate sufficient heme-bound soy leghemoglobin.
In addition, a modified transcription factor mxr1 gene (CS-mxr1-PICPA) under control of the P-aox1-PICPA promoter was introduced by transformation. The Mxr1 protein activates the P-aox1-PICPA promoter leading to increased expression of P-aox1-PICPA-driven CS-lgb2-SOYBN, heme biosynthesis genes, and CS-mxr1-PICPA itself.
Finally, the codon-optimized leghemoglobin gene from soybean (CS-lgb2-SOYBN) under the control of the P-aox1-PICPA promoter was introduced, leading to the expression of this gene in MXY0291.
According to the application documents (GRAS notice) the genome of MX0291 was completely sequenced and confirmed the presence of the following sequences:
- 16 copies of P-aox1-PICPA-CS-lgb2-SOYBN-T-aox1-PICPA
- 1 copy of P-aox1-PICPA-CS-mxr1-PICPA-T-fdh1-PICPA
- 1 copy of a portion of pMX349 - containing genes encoding heme biosynthesis enzymes 1 (CS-hem1-PICPA), 3 (CS-hem3-PICPA), 7 (CS-hem14-PICPA), and 8 (CS-hem15-PICPA) (no antibiotic resistance gene, no origin of replication)
- 2-3 copies of a portion of pMX346 - containing genes encoding heme biosynthesis enzymes 4 (CS-hem4-PICPA), 2 (CS-hem2-PICPA), 5 (CS-hem12-PICPA), and 6 (CS-hem13-PICPA) (no antibiotic resistance gene, no origin of replication)
There are indications that pMX346 copies were not fully integrated and portions of the CPG oxidase coding sequences (CS-hem13-PICPA) had been deleted in this strain (see Patent document).
The developer states that sequences are stably integrated and present in the strain over 150-200 generations of growth, which was confirmed by PCR. The final production strain does not contain any antibiotic resistance genes.
The purpose of the genetic modification is to obtain soy leghemoglobin by fermentation.
Following fermentation, cells are lysed and physically separated to remove insoluble materials and to concentrate soy leghemoglobin. The resulting liquid concentrate (LegH Prep) is a mixture containing soy leghemoglobin (6-9% w/w), residual P. pastoris proteins, and added stabilizers. The final product is heat treated to ensure the absence of viable production organism. Small amounts of residual P. pastoris DNA (about 0.2 mg/L) may also be present in the LegH Prep.
Impossible Foods developed soy leghemoglobin extracts (LegH Prep) as a heme-containing ingredient in meat-like products which imparts a meat-like flavor onto plant based food products. The first product containing LegH from P. pastoris to be commercialized in Australia, New Zealand, the U.S., Canada, Singapore, and Hong Kong was the Impossible Burger.
Sources:
GRAS Notice (GRN) No. 737
Patent US20170349906A1
Fraser et al. (2019), Safety Evaluation of Soy Leghemoglobin Protein Preparation Derived from Pichia pastoris, Intended for Use as a Flavor Catalyst in Plant-Based Meat. International Journal of Toxicology 37(3):241-262.